
Dye for materials
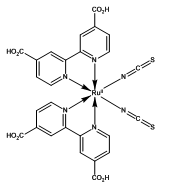
Ruthenium Dyes
In dye-sensitized solar cells, the dye is one of the key components for high power conversion efficiencies. In recent years, considerable developments have been made in the engineering of novel dye structures in order to enhance the performance of the system. In particular, amphiphilic homologues of the pioneering ruthenium based N-3 dye have been developed. These amphiphilic dyes display several advantages compared to the N-3 dye such as:
- a higher ground state pKa of the binding moiety thus increasing electrostatic binding onto the TiO2 surface at lower pH values,
- the decreased charge on the dye attenuating the electrostatic repulsion in between adsorbed dye units and thereby increasing the dye loading,
- increasing the stability of solar cells towards water-induced dye desorption,
- the oxidation potential of these complexes is cathodically shifted compared to that of the N-3 sensitizer, which increases the reversibility of the ruthenium III/II couple, leading to enhanced stability.
N-3
C26H16N6O8RuS2
Mol Wt: 705.64
N-719
C58H86N8O8RuS2
Mol Wt: 1188.55
Z-907
C42H52N6O4RuS2
Mol Wt: 870.10
Figure 2. Ruthenium-based N-3, N-719 and Z-907 dyes.
Table 1: Dyesol® dyes offered by Sigma-Aldrich Materials Science.
| Synonym | Product | Description | Product No. |
|---|---|---|---|
 N-3 N-3 |
cis-Bis(isothiocyanato) bis(2, 2’-bipyridyl-4, 4’-dicarboxylato ruthenium(II) | • Pioneering dye for use in DSSCs.
• Sensitizes wide band gap semi-conductors such as titanium oxide up to wavelengths of 700 nm. |
|
| N-719 | Di-tetrabutylammonium cis-bis(isothiocyanato)bis(2, 2’-bipyridyl-4, 4’-dicarboxylato)ruthenium(II) | • Modified dye to increase device voltage.
• Highly studied, high performance dye. |
|
| Z-907 | cis- | • Hydrophobic dye.
• Very efficiently sensitizes wide band-gap titanium oxide up to 750 nm. |
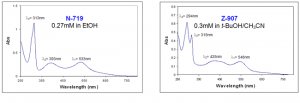
Ruthenium-based dyes exhibit ligand-centered charge transfer (LCCT) transitions (π - π*) as well as metal-to-ligand charge transfer (MLCT) transitions (4d - π*) that can be observed in the UV/Vis spectra of N-719 and Z-907 dyes (Figure 3). The absorption bands at lower energies represent the MLCT transitions (λ1 and λ2) whereas the more energetically demanding transitions (λ3 and λ4) correspond to LCCT transitions.
 Figure 3. UV/Vis spectra of N-719 (Product No. 703214) and Z-907 (Product No. 703168) dyes.
Figure 3. UV/Vis spectra of N-719 (Product No. 703214) and Z-907 (Product No. 703168) dyes.
A translucent DSC with an active area of 0.88 cm2 (8 mm x 11 mm) based on Dyesol test cell hardware prepared with N-719 dye is illustrated in Figure 4. The orange area is a result of the dye-modified titania; the yellow color is given by the electrolyte of the system.
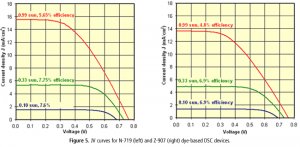
JV Curves
Standard test cells are characterised by typical JV plots as shown in Figure 5 when tested at three different sun levels (1 sun corresponds to 100 mW cm-2). The voltage, V, and the current density, J (mA cm-2), at the maximum power point are represented by Vmax and Jmax. Therefore the efficiencies can be calculated by:
η (%) = (Vmax x Jmax) / (sun level)
With commercially relevant low volatility electrolyte systems, typical short circuit currents are 15.5 mA cm-2 and 13.7 mA cm-2 at full sun for an efficiency of 5.7% and 4.8% for N-719 and Z-907 respectively. The efficiencies characteristically increase at lower sun levels in DSC systems, when compared to full sun level efficiencies, to 7.8% (5.2 mA cm-2) and 7.6% (1.5 mA cm-2) for N-719 and to 6.9% (4.8 mA cm-2) and 6.9% (1.4 mA cm-2) for Z-907 at 1/3 sun and 1/10 sun respectively.
Materials
References
- Chiba, Y.; Islam, A. ; Watanabe, Y. ; Komiya, R.; Koide, N. ; Han, L. Jpn. J. Appl. Phys. 2006, 45, L638.
- Grätzel, M. J. Photochem. Photobiol., A 2004, 164, 3.
- Chiba, Y.; Islam, A.; Kakutani, K. ; Komiya, R.; Koide, N.; Han, L. 15th International Photovoltaic Science & Engineering Conference (PVSEC-15), Shanghai, China, 2005, Technical Digest, p.665.
- Grätzel M. Inorg. Chem. 2005, 44, 6841.
- Klein, C.; Nazeeruddin, Md. K.; Di Censo, D.; Liska, P.; Grätzel, M. Inorg. Chem. 2004, 43, 4216.
- Hirata, N.; Lagref, J. J.; Palomares, E. J.; Durrant, J. R.; Nazeeruddin, Md. K. ; Grätzel, M.; Di Censo, D. Chem. Eur. J. 2004, 10, 595.

















